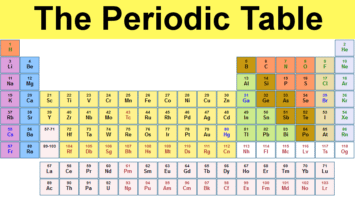
 Thus, the molar mass of bilirubin can be expressed as 584.73 g/mol, which is read as “five hundred eighty four point seventy three grams per mole. The division sign (/) implies “per,” and “1” is implied in the denominator. To find the molar mass, we need to add the atomic masses of two Hydrogen atoms and one Oxygen atoms. For example, the molar mass of Ba(OH) 2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: The molar mass of Ba(OH)2 requires the sum of 1 mass of Ba, 2 masses of O, and 2 masses of H: 1 Ba molar mass:īecause molar mass is defined as the mass for 1 mol of a substance, we can refer to molar mass as grams per mole (g/mol). Where more than one isotope exists, the value given is the abundance weighted average. For a fully interactive version with orbitals, isotopes, compounds, and free printouts, visit. This is approximately the sum of the number of protons and neutrons in the nucleus. Relative atomic mass The mass of an atom relative to that of carbon-12. The periodic table lists the atomic mass of carbon as 12.011 amu the average molar mass of carbonthe mass of 6. In formulas with polyatomic ions in parentheses, the subscript outside the parentheses is applied to every atom inside the parentheses. Density is the mass of a substance that would fill 1 cm 3 at room temperature. The molar mass of any substance is its atomic mass, molecular mass, or formula mass in grams per mole. The molar mass in grams is equal to the atomic weight of the element represented in atomic mass units. 
The atomic mass of an isotope and the relative isotopic mass.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
